by Matt Pettit, Chief Scientific Officer, Matcher Technologies Ltd
If IVF technologies have advanced so dramatically, is it acceptable that some safety practices have remained largely unchanged?
Since the late 1970s, assisted reproductive technologies have transformed the lives of millions. IVF has helped countless individuals and couples build families and preserve fertility for future family building, including many in same sex relationships. Outcomes have continued to improve, driven by advances such as vitrification for single embryo transfer, time lapse imaging, pre-implantation genetic testing (PGT), and most recently, the introduction of AI to support embryo selection.
But while these developments have improved success rates, they have also made IVF laboratories far more complex places to work. Each new technology requires additional steps. Each step requires verification. And the more steps there are, the more opportunities there are for something to go wrong.
Many of these extra tasks cannot yet be automated. They depend on the skill, dexterity, and concentration of embryologists. As a result, the workload has risen sharply. A biopsy cycle for PGT adds several time critical procedures to each embryo’s ICSI workflow. As a result, the number of identification checks required has almost doubled (estimated to be as many as 80 in some cases). Unless staffing levels grow at the same pace, which is often not possible due to the specialist skills required, clinics are exposed to higher levels of risk.
Global trends in IVF
At the same time, global demand for IVF continues to rise. Professional bodies such as SART, ESHRE and ASRM all report significant growth: treatment cycles have doubled in the past decade, the number of cryopreserved gametes and embryos per cycle continues to increase, cryostorage volumes and storage durations are expanding, and the use of PGT has grown worldwide. Births from donor sperm have tripled in some countries, while improved embryo selection and transfer practices have increased live birth rates while reducing multiple births.
More cycles, more donor material, longer storage, and more PGT all compound the workload, and with it, the risks of human error. Even the most skilled professionals are vulnerable as manual checks become increasingly inadequate. Mistakes may be relatively uncommon, but they are not rare. Recent media reports show they occur more often than many assume, and the ones that become public are merely the tip of the iceberg. In a field where a single error can have life‑changing consequences, the impact is profound and far reaching.
As an organisation that closely tracks developments in IVF quality control, we have observed a marked change in tone across the field in the last couple of years. Influential voices, along with insurers, legal stakeholders, hospital management and equity owners, are beginning to say, quite plainly, that enough is enough. The consensus is growing. Continuing to depend on manual processes alone is no longer defensible. Moving to a robust electronic witnessing and traceability system is no longer a ‘nice to have,’ it is essential.
Why IVF is more exposed to error today
Modern IVF places exceptional demands on embryologists. Selecting the best embryo for transfer is now central to improving success rates, especially in clinics committed to single embryo transfer. Technologies such as time lapse systems, PGT and AI may help identify the embryo with the highest potential, but they also increase the number of manual steps required to reach that decision and with it, the risks from mix-ups within a patient’s own embryos, as well as between different patients.
To carry out biopsy, vitrification and warming safely, staff must move gametes, embryos and biopsies through a series of tightly controlled procedures. Each move carries its own identification step. Each identification step must be correct and evidenced as correct. And as any human factors expert will confirm, the risk of error increases every time a person must perform a repetitive, high stakes task under time pressure.
On top of this, embryologists now bear responsibility for decisions that directly affect the long-term outcome of treatment. Selecting the wrong embryo could mean transferring one that has little chance of implanting or one that carries an inherited genetic variant. Or the devastating reality of an embryo being transferred to someone who is not its intended biological parent.
The emotional impact on the people affected is immense, and the professional weight carried by the teams involved is equally significant.
Human Factors such as stress, fatigue, pressure, distraction and complacency influence performance and error risk in every area of safety critical organisations. IVF is no different. Aviation refers to the phenomenon of drifting into unconscious routine as “involuntary automaticity”. In healthcare it is closely related to normalisation of deviance. Both create conditions where even experienced professionals can overlook hazards or skip steps without realising it.
Even the most conscientious humans will make mistakes. They always have. They always will. The only question is whether the system they work in is designed to catch those mistakes before they cause harm.
The IVF equivalent of Never Events
In wider healthcare, some mistakes are categorised as Never Events. These include wrong site operations, wrong transfusions, or catastrophic medication errors. They are not called Never Events because humans never err. They are called Never Events because they should never slip through the net – the system should prevent them.
IVF has its own equivalent. These include:
- transferring the wrong embryo
- mixing up samples
- mislabelling gametes, embryos or biopsies
- assigning the wrong PGT result to an embryo
- losing track of material in long-term cryostorage
- disposing of the wrong gametes or embryos
When these errors occur, the consequences are profound. There are ethical implications, emotional trauma, reputational damage, and often complex litigation that may even exceed the limits of a clinic’s insurance policy.
We often reassure ourselves that misidentification in IVF is extremely rare, but emerging evidence suggests the true rate may be higher than many assume. Studies that consider all categories of identification error, including those that are caught and corrected before reaching the patient, report manual misidentification rates of around 0.3 percent per procedural check. In a clinic performing 1000 cycles a year, with between 10 and 50 identification checks per cycle, this would translate to roughly 30 identification errors annually, even if most are intercepted before they cause harm. The important point is not the exact number. The important point is that these risks are predictable. And if they are predictable, they can be prevented.
Learning from other sectors
Other high-risk disciplines have already faced similar challenges. Aviation, pharmaceutical manufacturing, and blood transfusion services all recognised long ago that humans cannot guarantee perfect performance in complex environments. They responded by designing systems that compensate for human limitations.
Barcodes sit at the heart of many of these systems. In healthcare, barcodes are used to identify samples, devices, medicines, locations and even staff. The UK’s NHS Scan4Safety programme showed that using GS1 barcodes improved patient safety and saved millions of pounds. ISBT 128 barcoding transformed transfusion safety by ensuring that the right blood reaches the right patient every time.
These sectors accepted that manual checking alone cannot reliably prevent serious mistakes. IVF now faces the same reality.
Why barcodes are essential for IVF
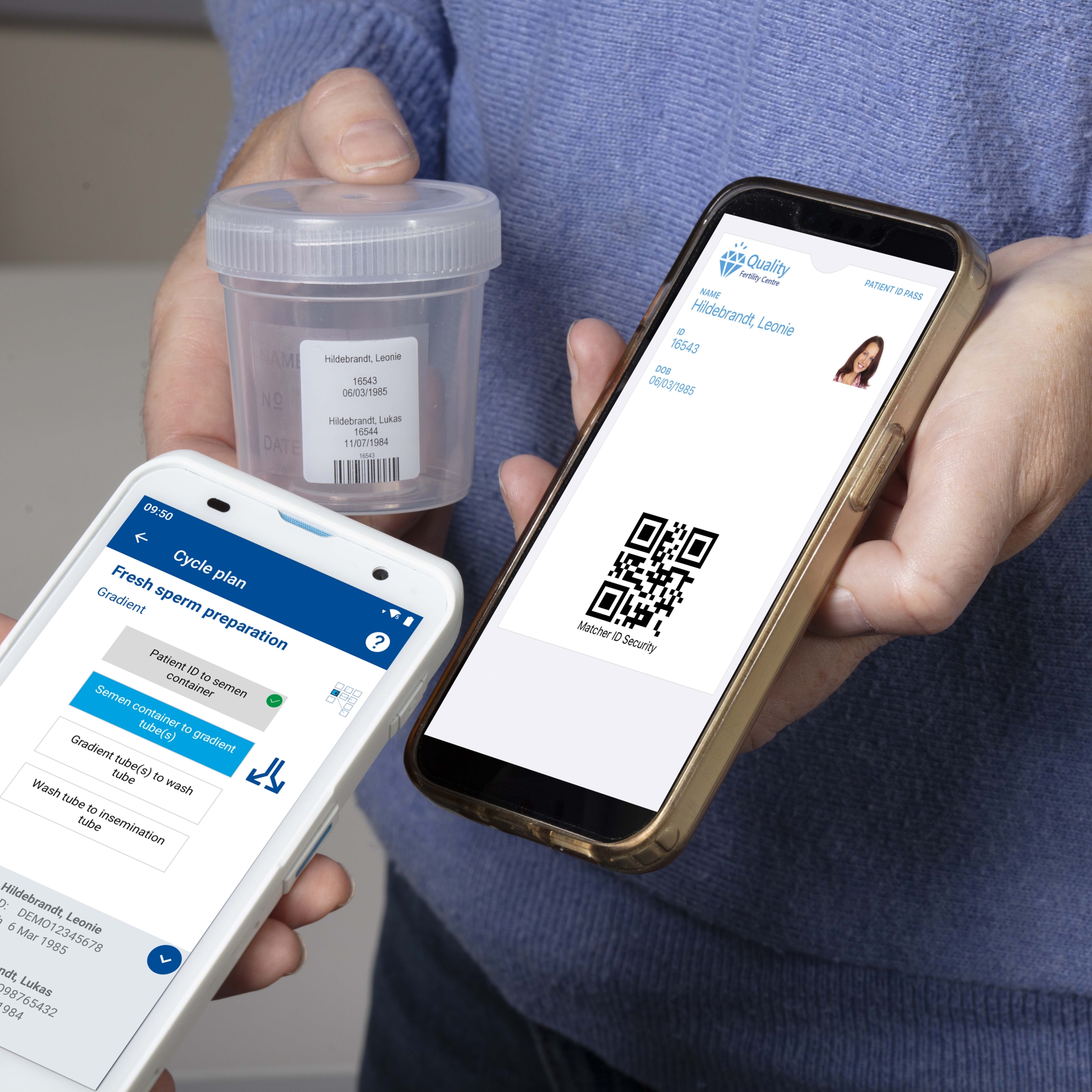
Barcodes offer a simple but powerful way to close the gap between manual and digital processes. They can provide globally unique identifiers that can be applied to patients, embryos, biopsy dishes, PCR tubes, vitrification devices, cryo locations and consumable products.
The benefits are clear.
Automated identification at every critical step
Barcode scanning can verify that the correct gametes and embryos are being used. If the wrong item is scanned, the system can alert the user immediately. This creates a forcing function that stops the process before harm can occur.
Embryo level tracking through the entire biopsy and vitrification workflow
A barcode placed on a culture well can be matched to correspondingly numbered biopsy dishes, PCR tubes and vitrification straws. This can ensure that the cells sent for PGT are from the same embryo that remains in storage. When the PGT results return, the link between embryo and outcome is already established.
Correct embryo selection before warming
Once results based on quality (and sometimes PGT) are known, the correct embryo must be selected for warming. This includes the right patient and the right embryo. Scanning the barcode provides confirmation. If the wrong embryo is chosen, a clear alert can signal the error instantly.
Even for embryos already in cryostorage that do not yet carry a barcode identifier, electronic tools are still available to provide the essential digital chain of custody. These systems allow clinics to document and verify each action, including capturing a photograph at the point of use, ensuring that even legacy material can be handled with greater confidence and accountability.
Seamless integration with other digital systems
Barcodes can link to EMRs, offsite biorepositories, genetics labs, satellite units, donor banks and even supply chains. They support a unified chain of custody that extends beyond the fertility centre.
Stronger quality management and photographic evidence
Systems such as Matcher can record every scan, every action, and even a photograph at each step. This creates a complete, time stamped, auditable history for every patient cycle.
This is more than witnessing. It is full traceability and complete accountability. That’s why the development of Matcher in partnership with Reprotech is centred on delivering true end‑to‑end traceability, with witnessing as just one element. It aims to provide a real‑time, multidirectional digital chain of custody that can extend across the entire ART ecosystem, recognising that chain of custody does not always begin or end within the four walls of a fertility center. And this level of assurance is only possible when systems connect and work together seamlessly to bridge the physical, procedural and communication gaps that pose a risk to patient and staff safety.
Why the old model cannot continue
Visual and verbal double witnessing served the profession for many years, but it has limits. It relies entirely on human memory and attention. It cannot provide photographic evidence, timestamped records or product level traceability. It cannot resist fatigue or distraction. It cannot integrate with digital systems. And it cannot offer the level of protection that modern IVF demands.
Expecting embryologists to deliver flawless performance under increasing pressure is unfair to them and unsafe for patients.
Other sectors have already accepted this truth and adapted accordingly. IVF must now do the same.
Conclusion: it is time for decisive change
Assisted reproduction is one of the most hopeful areas of medicine. But with hope comes responsibility. Clinics are entrusted with material that represents a family’s future. The systems that protect this material must be as advanced and robust as the technologies used to create embryos in the first place.
Preventable errors must become true Never Events in reproductive medicine.
Barcodes and digital traceability are essential to achieving this. They provide certainty, clarity, confidence and insights at every step, from registration to cryostorage to transfer. They support staff, protect patients and strengthen quality management.
The tools already exist. The risks are known. The benefits are clear.
Now is the moment for the IVF sector to take decisive action. The cost of doing nothing is simply too high.
References:
- Amagwula, T., Chang, P.L., Hossain, A., Tyner, J., Rivers, A.L. & Phelps, J.Y. (2012) ‘Preimplantation genetic diagnosis: a systematic review of litigation in the face of new technology’, Fertility and Sterility, 98(5), pp. 1277–1282.
- American Society for Reproductive Medicine (ASRM) (2007) ‘Cryopreservation’, Fertility and Sterility, 87(Suppl), pp. S23–S25.
- C.P. Moutos, R., Lahham, R. & Phelps, J.Y. (2019) ‘Cryostorage failures: a medicolegal review’, Journal of Assisted Reproduction and Genetics, 36, pp. 1041–1048.
- Ethics Committee of the American Society for Reproductive Medicine (2013) ‘Disposition of abandoned embryos: a committee opinion’, Fertility and Sterility, 99, pp. 1848–1849.
- ESHRE (2015) Revised guidelines for good practice in IVF laboratories. Available at: https://www.eshre.eu/Guidelines (Accessed: 23 October 2025).
- ESHRE PGT Consortium (various years) PGT Consortium Annual Reports.
- Forte, M. et al. (2016) ‘Electronic witness system in IVF – patients’ perspective’, Journal of Assisted Reproduction and Genetics, 33(9), pp. 1215–1222.
- Girnita, R.O. (2025) ‘Quantifying true error rate in assisted reproduction: a multi-site retrospective cohort analysis’, Middle East Fertility Society Journal, 30, p. 72.
- GS1 (2023) GS1 Healthcare recommendations on the use of GS1 standards to access digital content on medical products. GS1.
- HFEA (2024) Fertility Treatment 2023: Trends and Figures. London: Human Fertilisation and Embryology Authority.
- Holmes, R., Wirka, K.A., Catherino, A.B., Hayward, B. & Swain, J.E. (2021) ‘Comparison of electronic versus manual witnessing of procedures within the IVF laboratory: impact on timing and efficiency’, F&S Reports, 2(2), pp. 181–188.
- Human Fertilisation and Embryology Authority (2024) State of the Fertility Sector 2023–2024. London: HFEA.
- Ifenatuoha, C.W., Osakpolor, O. & Ajayi, A. (2023) ‘Errors in IVF laboratories: risk assessments and mitigations’, Middle East Fertility Society Journal, 28, p. 35.
- Jiang, M.X. et al. (2024) ‘IVF laboratory management through workflow-based RFID tag witnessing and real-time information entry’, Reproductive Biology and Endocrinology, 22, p. 96.
- Kimball Pomeroy, O., Comizzoli, P., Rushing, J.S., Lersten, I.L. & Nel-Themaat, L. (2022) ‘The ART of cryopreservation and its changing landscape’, Fertility and Sterility, 117, pp. 469–476.
- Narayan, S. et al. (2024) The 2024 Annual SHOT Report. Manchester: Serious Hazards of Transfusion (SHOT) Steering Group.
- Nemec, E.C. (2011) ‘ASHP Statement on BarCode Verification During Inventory, Preparation, and Dispensing of Medications’, American Journal of HealthSystem Pharmacy, 68(5), pp. 442–445.Code Verification During Inventory, Preparation, and Dispensing of Medications’, American Journal of HealthSystem Pharmacy, 68(5), pp. 442–445.
- Rich Vaughn (2017) Embryo disposition and cryopreservation legal issues. American Bar Association / International Fertility Law Group.
- Scan4Safety (NHS) (no date) Scan4Safety. Available at: https://scan4safety.nhs.uk/
- Society for Assisted Reproductive Technology (SART) (various years) SART Annual Reports.
- Thornhill, A.R. et al. (2011) ‘Reducing human error in IVF with electronic witnessing’, Fertility and Sterility, 96(3 Suppl), p. S179.
- Thornhill, A.R., Orriols Brunetti, X. & Bird, S. (2013) ‘Measuring human error in the IVF laboratory using an electronic witnessing system (RI Witness)’, 17th World Congress on Controversies in Obstetrics, Gynaecology & Infertility (COGI), Lisbon.
- U.S. Food and Drug Administration (FDA) (2022) Unique Device Identification System (UDI System).






Contact
Matcher Technologies Ltd
1b Canalside Business Park
Tattenhall
Chester
CH3 9BD
United Kingdom
Tel: +44(0)1829 771 327
Email: info@imtmatcher.com
Additional Information
Copyright
Copyright © 2002-2025 Matcher Technologies Ltd.
